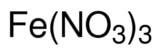| naming_booklet_-_large_with_answers.doc | |
| File Size: | 402 kb |
| File Type: | doc |
Periodic Table Trends and Reactivity
What causes elements to combine to form compounds? Why are some elements so reactive compared to others?
Electrons are the answer! In particular, those electrons we find on the outer most shell of an atom, the valence electrons. Every atom wants to be like their nearest noble gas, that is, they want to have the same electron configuration. Why do they want to be like them? It's not because they are "noble" and rule the other elements.... it's because they have a full outer shell of electrons, which makes them stable!
It turns out that elements that have the same number of valence electrons have similar chemical properties, which is why they are grouped together into "families".
Electrons are the answer! In particular, those electrons we find on the outer most shell of an atom, the valence electrons. Every atom wants to be like their nearest noble gas, that is, they want to have the same electron configuration. Why do they want to be like them? It's not because they are "noble" and rule the other elements.... it's because they have a full outer shell of electrons, which makes them stable!
It turns out that elements that have the same number of valence electrons have similar chemical properties, which is why they are grouped together into "families".
| families_and_reactivity.pptx | |
| File Size: | 460 kb |
| File Type: | pptx |
Ions
When elements bond to other elements, they do so to have a full outer shell of electrons. With Ionic compounds some elements will gain electrons to get there, and others will lose electrons.
Knowing how each element fills up it's outer most shell will tell you the charge the atom will take in an ionic bond.
Knowing how each element fills up it's outer most shell will tell you the charge the atom will take in an ionic bond.
Ionic Bonding
An ionic bond is a type of chemical bond that involves the electrostatic attraction between oppositely charged ions. Ions are atoms that have lost one or more electrons (known as cations) and atoms that have gained one or more electrons (known as anions).
Naming Binary Ionic Compounds and Ionic Formulas
Naming binary ionic compounds follows only two rules:
1) do not change the name of the cation
2) add the suffix "ide" to the anion. for example oxygen becomes oxide
Writing Ionic formulas is more complicated and involved taking into consideration how many electrons each ion wants to lose/gain. The key to determining an ionic formula is ensuring that the number of electrons lost is the same as the number of electrons gained (the charges "balance")
1) do not change the name of the cation
2) add the suffix "ide" to the anion. for example oxygen becomes oxide
Writing Ionic formulas is more complicated and involved taking into consideration how many electrons each ion wants to lose/gain. The key to determining an ionic formula is ensuring that the number of electrons lost is the same as the number of electrons gained (the charges "balance")
Ionic bonding - Multivalent Metals
Most cations in ionic compounds will always have the same oxidation state, that is, they will always lose the same number of electrons. However, the transition metals are unique in that they have multiple oxidation states.
Alfred stock realized that there will be a fundamental problem when naming compounds containing the transition metals, so he devised a system using roman numerals to represent the charge of the transition metal ion.
Alfred stock realized that there will be a fundamental problem when naming compounds containing the transition metals, so he devised a system using roman numerals to represent the charge of the transition metal ion.
Polyatomic Ions
Polyatomic ions are exactly as they sound. It is a charged ion composed of two or more atoms covalently bonded together.
Note that we treat the polyatomic ion as a single unit. Thus, when we have more than one of a polyatomic ion in an ionic bond we must put our polyatomic ion in brackets. For example:
Note that we treat the polyatomic ion as a single unit. Thus, when we have more than one of a polyatomic ion in an ionic bond we must put our polyatomic ion in brackets. For example:
In this example of Iron (III) Nitrate, there are three nitrates. Another rule when dealing with polyatomic ions is that you do not change their name when naming an ionic compound.
Molecular Compounds (AKA Covalent)
A covalent bond is the sharing of valence electrons between two non-metals in order to achieve a full outer shell of electros. Unlike ionic bonds, it is the attraction between electrons and the opposing atoms nuclei which create these bonds.
Remember it is important to learn your greek prefixes in order to accurately describe covalent compounds.
Remember it is important to learn your greek prefixes in order to accurately describe covalent compounds.
The Law of Conservation of Mass
Matter cannot be created or destroyed in a chemical reaction.
This is another one of the big guiding principles in chemistry. For our purposes it means that the number and type of atoms in the reactants will be the same in the products. And since the atoms mass doesn't change in a chemical reaction, the mass of the reactants will also be equal to the mass of the products.
This is the reason why we must "balance" chemical reactions. The below video shows conservation of mass in action (note that after 2:50 it is no longer talking about chemical reactions)
This is another one of the big guiding principles in chemistry. For our purposes it means that the number and type of atoms in the reactants will be the same in the products. And since the atoms mass doesn't change in a chemical reaction, the mass of the reactants will also be equal to the mass of the products.
This is the reason why we must "balance" chemical reactions. The below video shows conservation of mass in action (note that after 2:50 it is no longer talking about chemical reactions)
Word equations and Chemical Equations
Chemical reactions in this course will be illustrated in two different ways. With either a word equation or a chemical equation. Converting between word and chemical equations is common practice in chemistry, and will really test your understanding of bonding. Having the correct number and type of atoms in your chemical reaction is important to properly illustrate conservation of mass.
If you are struggling coming up with the Skeleton equation, you need to review ionic and covalent bonding.
If you are struggling coming up with the Skeleton equation, you need to review ionic and covalent bonding.
Balancing Chemical Equations
Balancing chemical equations is like coming up with a recipe for your reaction. It tells you how many of each element and/or compound react to form your products.
Types of Reactions
Although not covering all types of reactions (you are going to learn about organic reactions and redox reactions), you now know about five major types of reactions. Understanding these five types of reactions will allow you to predict products given reactants.
| 13_-_reaction_types__notes_.docx | |
| File Size: | 26 kb |
| File Type: | docx |
Acids and Bases
Our evolution of taste buds for sour and bitter may have saved our ancestors from spoiled foods and dangerous poisons, but today we can use them to identify acids and bases!
Acids and bases are a part of our everyday lives and are an important part of understanding chemistry.
Acids and bases are a part of our everyday lives and are an important part of understanding chemistry.
| 12_-_balancing_equations__practice_key_.pdf | |
| File Size: | 67 kb |
| File Type: | |
| 13_-_synthesis___decomposition_reactions__practice_key_.pdf | |
| File Size: | 50 kb |
| File Type: | |
| 14_-_single___double_replacement__practice_key_.pdf | |
| File Size: | 61 kb |
| File Type: | |
| 15_-_combustion_reactions__practice_key_.pdf | |
| File Size: | 22 kb |
| File Type: | |
| balance_worksheets__lots__-_answers.docx | |
| File Size: | 258 kb |
| File Type: | docx |