| 04_all_stoich_practice_sheets.docx | |
| File Size: | 42 kb |
| File Type: | docx |
| chem_30s_-_extra_stoicheometry_practice_with_answers.docx | |
| File Size: | 71 kb |
| File Type: | docx |
| chem_30s_-_arial_assassination_-_fall_2019.docx | |
| File Size: | 36 kb |
| File Type: | docx |
| chem_30s_-_two_tickets_for_styx_-_fall_2019.docx | |
| File Size: | 68 kb |
| File Type: | docx |
Isotopes & Average Atomic Mass
Why is chlorine's atomic mass 35.45 atomic mass units?
You learnt in previous years that we should round this number off when calculating the number of neutrons... which would give us 17 protons and 18 neutrons. However 17 electrons weigh an insignificant amount and doesn't account for the nearly half an amu "extra" above 35.
The answer is isotopes, atoms of the same element with differing numbers of neutrons. In nature chlorine has two stable isotopes, chlorine-35 (18 neutrons) and chlorine-37 (20 neutrons).
The number represented on your periodic table is the average atomic mass based on the relative abundance of that elements isotopes.
You learnt in previous years that we should round this number off when calculating the number of neutrons... which would give us 17 protons and 18 neutrons. However 17 electrons weigh an insignificant amount and doesn't account for the nearly half an amu "extra" above 35.
The answer is isotopes, atoms of the same element with differing numbers of neutrons. In nature chlorine has two stable isotopes, chlorine-35 (18 neutrons) and chlorine-37 (20 neutrons).
The number represented on your periodic table is the average atomic mass based on the relative abundance of that elements isotopes.
Stoichiometry
Studies the amount of substances that are involved in reactions. Stoichiometry is effectively the language of chemistry. Chemists use this language ALL THE TIME, as you will be as we become chemists this semester!
Scientific Notation
Imagine how much of a pain it would be in order to do mathematical manipulations with exceptionally large numbers and exceptionally small numbers. For example:
(602000000000000000000000) * (0.0000000000000786)
It would be so easy to miss a zero when entering this into your calculator, and time consuming to boot. Now consider these numbers represented using scientific notation.
(602000000000000000000000) * (0.0000000000000786)
It would be so easy to miss a zero when entering this into your calculator, and time consuming to boot. Now consider these numbers represented using scientific notation.
(6.02 x 1023) * (7.86 x 1014)
The Mole
As humans, we work on a macroscopic scale. It is challenging for us to even imagine things at the atomic level, never mind work with particles at that level.
As a result scientists created the mole, the "measuring cup" of chemistry. In one mole there are 602 sextillion particles. An amount that allows us to do measurements of chemical reactions on a scale that we can relate to and observe.
As a result scientists created the mole, the "measuring cup" of chemistry. In one mole there are 602 sextillion particles. An amount that allows us to do measurements of chemical reactions on a scale that we can relate to and observe.
Particles to Moles and Moles to Particles
Converting between the number of particles to moles and vise versa is an important skill. Since atoms are so small we need to convert them into a amount that we can relate to. Learning to use this "measuring cup" is essential to learning the language of chemistry.
| 04_moles_to_particles.docx | |
| File Size: | 28 kb |
| File Type: | docx |
Moles to Mass and Mass to Moles
The mole is defined as 12 grams of carbon-12. Since the amu is also defined based on carbon-12, this allows us to convert between moles and grams of a substance by using the molar mass.
| 05_moles_to_mass.docx | |
| File Size: | 28 kb |
| File Type: | docx |
Mass to Particles and Particles to Mass
Dimensional analysis is key to solving complex stoichiometric problems. Converting particles <--> grams will utilize both conversion factors we have learned about, avagadro's number and molar mass.
| 06_mass_to_particles.docx | |
| File Size: | 28 kb |
| File Type: | docx |
Moles to Liters of Gas and Liters of Gas to Moles
Amedeo Avagadro was the first to postulate that the same volume of two gases at the same temperature and pressure would contain the same number of particles. In practical terms, the volume of 1 mole of gas at standard temperature and pressure takes up a volume of 22.4 L.
Standard temperature and pressure --> 0 degrees Celsius and 1 atmosphere of pressure.
Standard temperature and pressure --> 0 degrees Celsius and 1 atmosphere of pressure.
| 07_moles_to_liters.docx | |
| File Size: | 27 kb |
| File Type: | docx |
| 08_mole_conversions.docx | |
| File Size: | 30 kb |
| File Type: | docx |
Percent Composition
Determining the percent composition by mass, while seemingly a pointless skill will play a particular importance when determining the empirical and molecular formula of a compound.
% composition = (mass of the element / mass of the compound) * 100
% composition = (mass of the element / mass of the compound) * 100
Empirical Formula and Molecular Formula
The empirical formula is the LOWEST whole number ratio of elements in a compound.
The molecular formula represents how many of each type of atom are in the compound. ie. it is the "regular formula"
For example, glucose has a molecular formula of C6H12O6 and a empirical formula of CH2O
The molecular formula represents how many of each type of atom are in the compound. ie. it is the "regular formula"
For example, glucose has a molecular formula of C6H12O6 and a empirical formula of CH2O
| 09_empirical_and_molecular_formula.docx | |
| File Size: | 28 kb |
| File Type: | docx |
Formula of a Hydrate
Hydrates (inorganic) are ionic solids which have water "tied up" in it's crystal lattice. This water is bound by polar interactions between water and the ions in the compound.
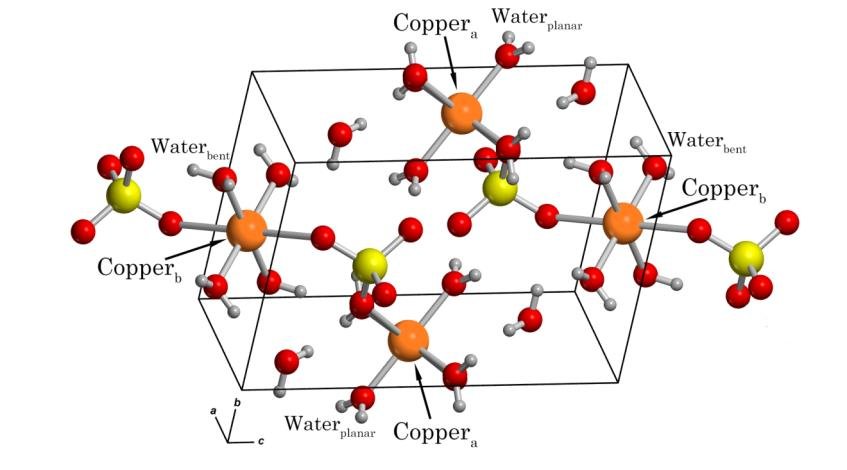
These complexes will have a specific unchanging ratio of water molecules to formula units of the compound. The image below shows the structure of copper (II) sulfate pentahydrate
These complexes will have a specific unchanging ratio of water molecules to formula units of the compound. The image below shows the structure of copper (II) sulfate pentahydrate
| hydrates_practice.pdf | |
| File Size: | 15 kb |
| File Type: | |
Mole to Mole Stoichiometry
When trying to assess the quantity of a reactant that is consumed, or a product that is produced, the mole ratio as described by the balanced chemical equation must be used. Remember the balanced chemical equation is your "recipe" for the reaction!
| 10_mole_to_mole_stoich.docx | |
| File Size: | 29 kb |
| File Type: | docx |
Stoichiometric Conversions
When doing stoichiometric problems you often will be given amounts of substances that first must be converted into moles. Remember that moles is the universal language through which we talk about quantities in our reaction (our balanced chemical equation).
When solving any stoichiometric problem you should follow the following steps:
1) Create a balanced chemical equation
2) If your given quantity isn't in moles, convert to moles
3) Using the mole ratio, find the moles of your unknown (what the question asks for)
4) If the question asks for an amount that isn't moles, convert the quantity of your unknown.
When solving any stoichiometric problem you should follow the following steps:
1) Create a balanced chemical equation
2) If your given quantity isn't in moles, convert to moles
3) Using the mole ratio, find the moles of your unknown (what the question asks for)
4) If the question asks for an amount that isn't moles, convert the quantity of your unknown.
| 11_mixed_stoich_problems.docx | |
| File Size: | 33 kb |
| File Type: | docx |
Limiting Reagent
Up to this point we have only explored reactions in which you were told which reactant was limiting and which was in excess. However, determining which reactant is in fact the limiting reagent to a reaction is an important skill. The limiting reagent determines how much product can be formed from the reaction as it will be the one to run out first.
This is a challenging skill that will require lots of practice!
This is a challenging skill that will require lots of practice!
| 12_limiting_reagent_problems.docx | |
| File Size: | 30 kb |
| File Type: | docx |
More Stoich Practice
The key to mastering any skill is practice !
| stoich_mole_to_mole_worksheet.pdf | |
| File Size: | 276 kb |
| File Type: | |
| stoich_mole_to_mole_worksheet_answers.pdf | |
| File Size: | 147 kb |
| File Type: | |
| stoich_mole_to_mass_worksheet.pdf | |
| File Size: | 337 kb |
| File Type: | |
| stoich_mole_to_mass_worksheet_answers.pdf | |
| File Size: | 507 kb |
| File Type: | |
| stoich_mole_and_mass_mixed_worksheet.pdf | |
| File Size: | 301 kb |
| File Type: | |
| stoich_mole_and_mass_mixed_worksheet_answers.pdf | |
| File Size: | 485 kb |
| File Type: | |
| stoich_mass_to_mass_worksheet.pdf | |
| File Size: | 330 kb |
| File Type: | |
| stoich_mass_to_mass_worksheet_answers.pdf | |
| File Size: | 484 kb |
| File Type: | |
| stoich_limiting_reagent_worksheet.pdf | |
| File Size: | 143 kb |
| File Type: | |
| stoich_limiting_reagent_worksheet_answer.pdf | |
| File Size: | 207 kb |
| File Type: | |
| limiting_reagent_worksheet.docx | |
| File Size: | 14 kb |
| File Type: | docx |
| limiting_reagent_worksheet_answers.pdf | |
| File Size: | 284 kb |
| File Type: | |